Are Noble gasses truly inert?
- kieronconway
- Dec 22, 2025
- 6 min read

When writing about noble gases in a Journey into Modern Physics, the expression ' react with nothing, under normal conditions' was always used. This article explores noble gas chemistry, which may come as a surprise. Under certain conditions, some of the noble gasses do react with other atoms, in particular the more massive gasses are more reactive than the lighter ones as their outermost orbitals are not as strongly attached to the nucleus. However, we are not talking about the classic covalent and ionic bonds of chapter 4 – it's far more exotic!
What makes an atom a noble gas is the fact that in their outermost orbitals, they have a full, spherical, 's' type orbital and three full 'p' type orbitals, each having a two lobed shape, orientated along different axes. Each lobe can only be occupied by two electrons with opposite spin orientations. This configuration makes them very reluctant to react with other atoms in the normal manner;
Their valence shell is full (the s and three p orbitals)
They have very high ionisation energies (the energy required to knock one of their electrons out of the atom and set it free).
They have very low electron affinity (they don't want any other atom's valence electrons).
From chapter 4 in Part 1 of a Journey into Modern Physics, the outermost shell of all noble gasses are defined as sN(2), pN(6) were 's' and 'p' are the designations of the orbitals involved in a noble gas's outer shell and the N defines which shell, from 2 onwards. The number in brackets indicates the total number of electrons accommodated. The 's' type orbital has a hollow, spherical shape and the 'p' orbitals consist of three orbitals, each orientated in a different direction. These orbitals are illustrated below.

The noble gasses have the following outer shell configurations;
Helium: s1(2) there are no p2 orbitals in helium
Neon: s2(2), p2(6)
Argon: s3(2), p3(6)
Krypton: s4(2), p4(6)
Xenon: s5(2), p5(6)
Radon: s6(2), p6(6) radioactive
Oganesson; s7(2), p7(6) highly unstable and not found in nature
First up - helium
Helium has the highest ionisation energy, you just can't normally separate the helium nucleus from its two electrons and it has no affinity for electrons. As a result, helium really is inert in that there is no chemistry involved with helium. No conventional chemical compounds involving helium exist under any conditions. You can produce helium ions and you can excite its electrons, but that's about it.
Unless...... it finds itself in a very high pressure environment. By high pressure we are talking more than 1 million atmospheres and sodium helide has been observed. It's not a chemical bonding between sodium (one electron in its valence shell) and helium, but helium under these absurd pressures is pushed into sites within the sodium's lattice structure. You could call it a 'space-filling, electron-repelling parasite' at ultra-high pressure!
Next up: neon.
No stable neon compounds have ever been observed, it's as inert as helium. Even at very high pressures, no stable compounds have been confirmed. So neon is king of the inert noble gasses at present.
Next up: argon.
Under normal conditions, argon is inert, but at a few degrees above absolute 0, it has been made to form genuine chemical bonds in a compound called argon fluoride with a chemical formula HArF. Fluorine is an exceptionally strong electron acceptor, consisting of one full s2 orbital, two full p2 orbitals and one partially empty p2 orbital in its outermost shell, which has a total of 7 electrons.
The bonding is best described as highly-polar covalent, often represented as HArF with strong charge separation. The fluorine draws the electrons of the Argon towards it creating a slight positive charge on the Argon. The Hydrogen then binds to the Argon in a weak form of electron sharing. The molecule is very unstable and breaks apart if the temperature rises above 40 degrees Kelvin. Despite its fragility, HArF is regarded as a genuine compound.
Next up: krypton
There are a number of compounds that can be created with krypton, but ultra-low temperatures are involved. The most well-known compound is krypton difluoride where fluorine's ability to attract electrons distorts the electron clouds in the outer shell of krypton.
Because of krypton's size, its outer electrons are not held as strongly as those of the lighter noble gasses and are more easily attracted to fluorine.
Imagine krypton difluoride, depicted as F-Kr-F, instead of having two normal bonds, three atoms share four electrons. The resultant molecule has a linear bonding axis represented by F-Kr-F. The bond is best described as the single p orbital in Kr, with two electrons, lying along the axis of the molecule being shared with one partially populated p orbital from each fluorine atom, each containing 1 electron. The result is three atomic orbitals are made to overlap and are converted to three molecular orbitals in a quantum bond, sharing four electrons, which is best described using molecular orbital theory.
The bonding possess both covalent and ionic character, creating a relatively stable molecule at low temperatures. It is a weak bond and can easily be broken, especially if the temperature rises.
F-Kr-F is all about fluorine's ability to pull electrons from Kr towards it producing lower energy configurations that create a genuine, if fragile, molecular bonding structure.
Fluorine showed that it is possible to form bonds that are based on quantum mechanics.
Next up: xenon
In 1962, Neil Bartlett created the first noble gas compound using xenon. Up to this time, column 18 elements were referred to as 'inert elements', but as this no longer appeared to be the case, despite the exotic quantum nature of molecular bonding, their title was demoted to 'noble gasses'.
In fact, Xenon is the most chemically reactive of all the noble gasses. It can form fluorine compounds, which are stable enough to be studied in labs. There are also oxides of Xenon, which together with the fluorine compounds exhibit shared electron density in the form of actual orbitals rather than individual electrons. Compounds are;
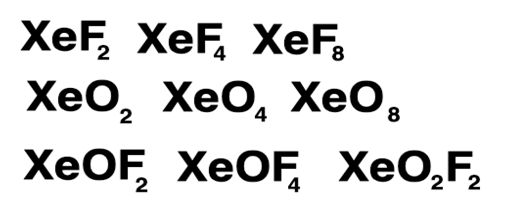
Just remember that the reason that these bonds come into existence is that they create a low energy configuration for the molecular structures formed without the need for covalent or ionic bonds, which the noble gases don't engage in. This chemistry is all about quantum bonding in molecular structures. Xenon is the most chemically active of all the noble gases as its outer electron orbitals are weaker than the lighter noble gasses.
Next up radon
Radon is a naturally occurring gas produced in the decay chain of uranium and is itself radioactive decaying through alpha emission into polonium, which continues the decay chain all the way to stable lead. As it only has a half-life of just under 4 days, it is not a good subject for laboratory research and may well be even more chemically active than xenon.
Then there's oganesson
Oganesson is the heaviest and last element in the periodic table, synthesised by Yuri Oganessian in 2002. It is the last of the super-heavy elements in the table. It's so unstable, that it's incredibly difficult to study. At the time of writing it holds the record for having the highest atomic number, 118 and mass in the periodic table. Apparently, only a handful of atoms of oganesson have ever been synthesised and they have a half-life of just a few milliseconds.
To produce Oganesson, you need to bombard californium (Cf) with calcium (Ca) ions in an accelerator and hope that they merge to produce oganesson and turf out three neutrons from the final product. The reaction is;

Believe it or not, you have to run the beam of calcium ions for hundreds of hours to produce a single atom! As you can imagine, it's very difficult to study to see if it engages in any form of chemistry.
Conclusion
Under normal conditions, the noble gases are inert. Under extreme conditions of high pressure or low temperature, the noble gasses can and do form molecules with other atoms through sharing of electron density without the need to disrupt the s or p electron content.
This article is all about physics explaining chemistry interactions.
© 2025 Kieron Conway - All rights reserved.
--
Liked this article? Check out:
where you can read all about an exciting new science series: A Journey into Modern Physics, available in three parts, from Amazon on-line shops. (Some of the above article may appear in this series).
Also, the web-site has an index of blog articles published to date, for easy access to an article that might interest you.
--



Comments